Olaprixa Industrial
Olaprixa Industrial












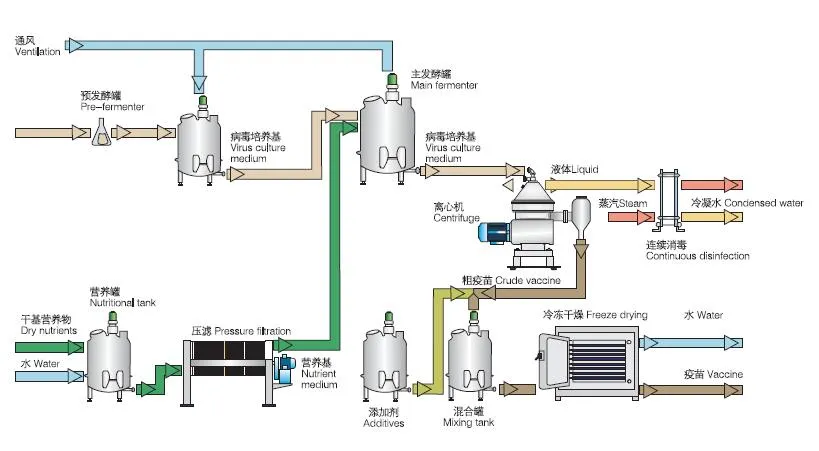
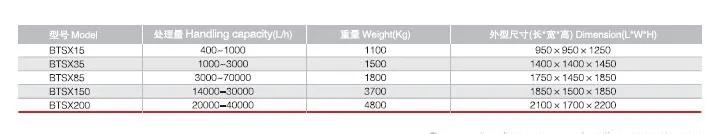
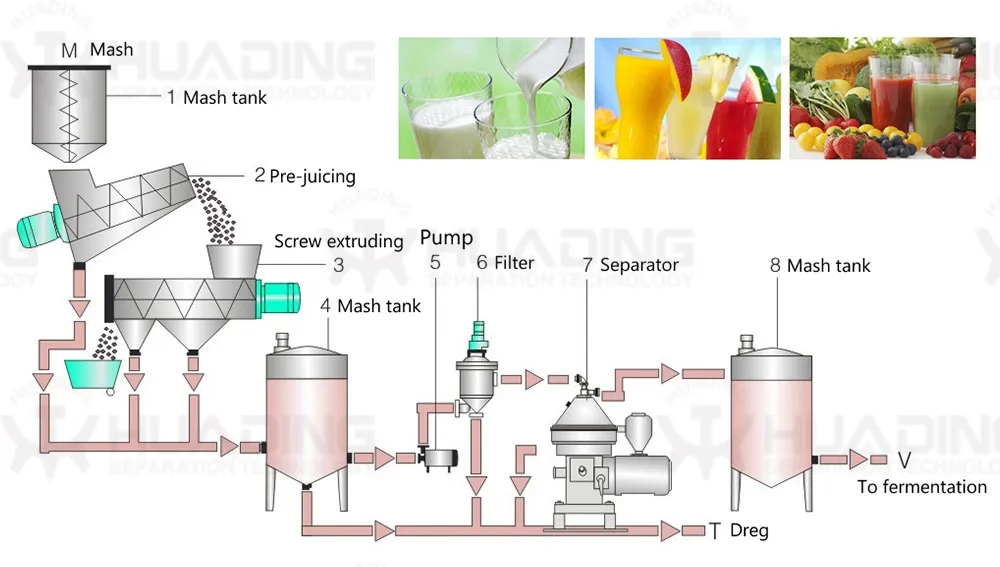
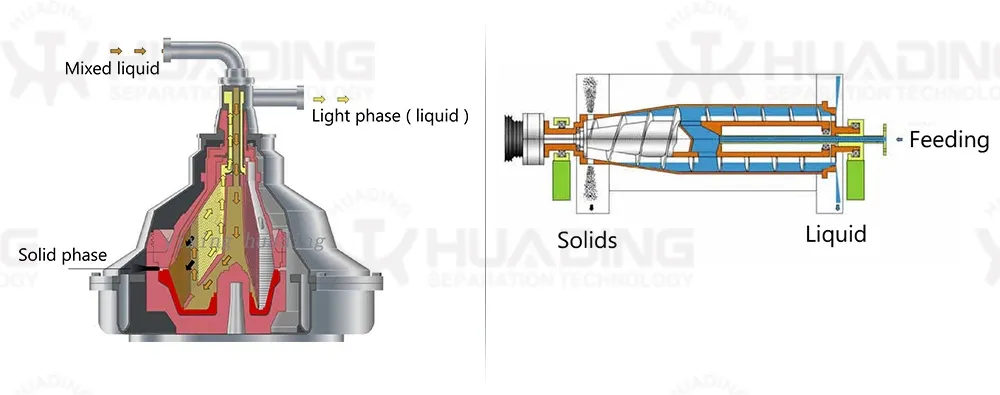
Centrifugal separation method is an important part of many processes in pharmaceutical biotechnology. The quality of the separating process is decisive to the quality of the end product and the economy and environmental compatibility of the process. The constantly high separating rates of separators and decanters comply fully with these requirements.
The wide product range has realized the application of separators and decanters in different fields as well as application in all processes requiring phase separation technology. Our centrifugal separation technology plays a key role in biotech and pharmaceutical processes.
These systems are in compliance with production requirements of aseptic process management, optimum cleaning capability, closed product handling, gentle product treatment, reliable compliance with GMP requirements, and efficient recovery of active ingredients.