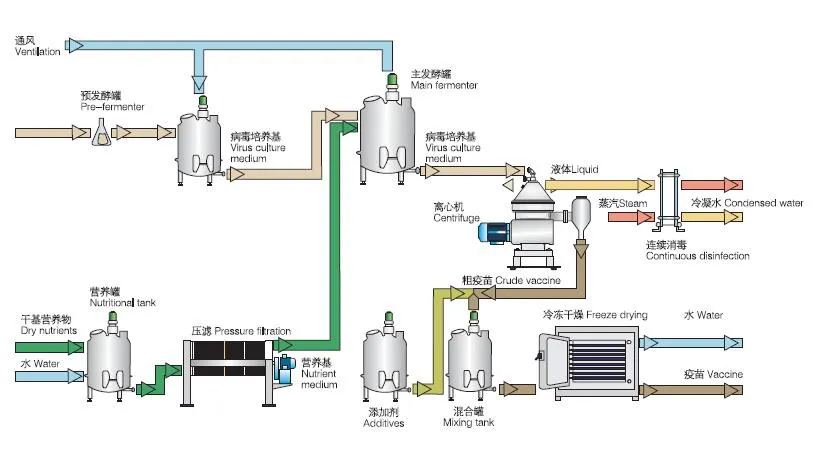
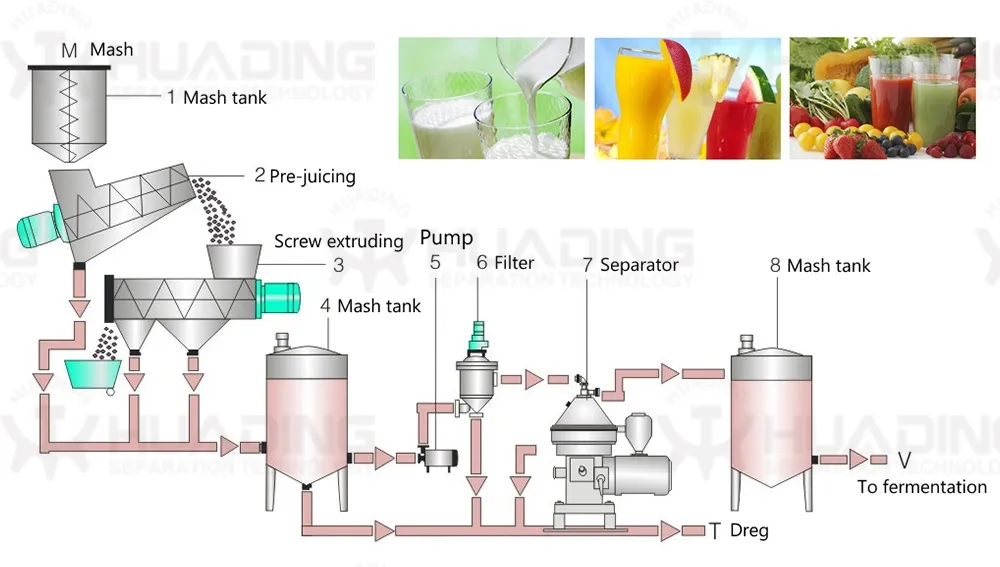
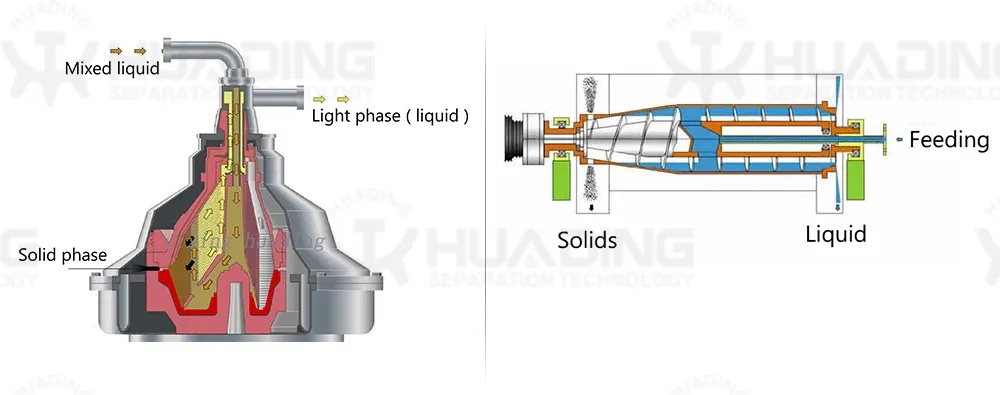
Centrifugal separation method is an important part of many processes in pharmaceutical biotechnology. The quality of the separating process is decisive to the quality of the end product and the economy and environmental compatibility of the process. The constantly high separating rates of separators and decanters comply fully with these requirements.
The wide product range has realized the application of separators and decanters in different fields as well as application in all processes requiring phase separation technology. Our centrifugal separation technology plays a key role in biotech and pharmaceutical processes.
Separators and decanters are in compliance with production requirements of aseptic process management, optimum cleaning capability, closed product handling, gentle product treatment, reliable compliance with GMP requirements, and efficient recovery of active ingredients.
Industrial Applications
Antibiotics
Alkaloids
Flavourings
Perfumes
Enzymes
Plant extracts
Organ extracts
Glucose isomerase
Hormones
Human blood
Vaccines
Medicinal plants
Rose oils
Synthetic pharmaceutical products
Vitamins
Fermentation products
Frequently Asked Questions
What are the primary materials used in the BTSX centrifuge?
The system is constructed using high-grade stainless steel options including SS304, 316L, and 2205 to ensure durability and chemical resistance in pharmaceutical environments.
Is this equipment compliant with GMP requirements?
Yes, the separators are designed for aseptic process management, featuring optimum cleaning capability and closed product handling to meet strict GMP standards.
What type of control system does the centrifuge use?
The equipment is equipped with a high-quality Siemens HMI (Human Machine Interface) for precise and reliable operation control.
Which industries can benefit from this separation technology?
It is widely applicable in pharmaceutical biotechnology, food and beverage factories, farms, energy, mining, and sludge treatment industries.
What kind of after-sales support is available?
We provide comprehensive support including field installation, commissioning, training, online technical assistance, and video support.
What is the production capacity for this model?
The production capacity is approximately 500 sets per year, ensuring steady availability for large-scale industrial needs.





 Olaprixa Industrial
Olaprixa Industrial